Leafy Greens: Keeping Salad Favorites Safe To Eat
|
|
In the 20 years or so since packaged salad mixes first began showing up in supermarkets nationwide, we’ve made them a produce-section favorite. It’s no wonder. These bagged mixes—washed, cut, and ready to enjoy—offer convenience, selection, and quality, and perhaps best of all, they free us from the chore of washing and chopping, slicing, or shredding salad veggies.
But outbreaks of foodborne illness have, from time to time, been associated with bagged salad greens. The outbreaks have led the fresh-cut produce industry to voluntarily adopt stringent quality-control standards. The standards help ensure the safety of dozens of different kinds of salad staples, from iceberg and romaine lettuces to spinach, radicchio, and many more.
Helping growers and processors keep these fresh-cut veggies safe to eat is a priority of Agricultural Research Service food safety researchers, including scientists in the Produce Safety and Microbiology Research Unit. The team is part of the agency’s Western Regional Research Center in Albany, California, in the San Francisco Bay area.
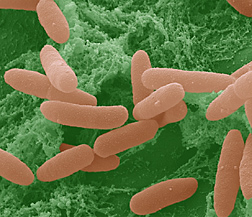
Innovative studies led by ARS microbiologist Maria T. Brandl are providing new information about the impressive array of genes that Escherichia coli O157:H7 calls into action when attempting to colonize leaves of fresh-cut lettuce. In such situations, the pathogenic microbe is essentially trying to stay alive while surrounded by natural chemicals leaking from broken lettuce cells.
|
|
Cells Get Sliced, Too
Mechanical cutting of lettuce leaves into large pieces or shredding of leaves into narrow strips, like those in taco filling, breaks lettuce cells. Lettuce cells can also be injured if leaves are bruised—during harvest or while at the processing plant, for instance.
For E. coli, the good news is that broken lettuce cells exude carbohydrates, which the microbe can use as a source of energy. But the bad news, from the microbe’s point of view, is that injured leaf cells can also leak compounds that are problematic for the pathogen.
Oxidants are a good case in point. Wounded lettuce cells may give off a burst of hydrogen peroxide, for example, an oxidant that can, as its name suggests, cause oxidative stress for E. coli.
The pathogen’s response to oxidative stress is one example of a coping strategy that’s of keen interest to Brandl and her colleagues. “Chlorine, the most widely used sanitizer in produce processing, is an oxidant,” says Brandl. “Our findings suggest that E. coli cells that have already encountered oxidative stress imposed by plant-cell oxidants, and have activated genes to overcome that stress, may be better adapted to withstand chlorine sanitizers during washing and processing than E. coli cells that have not been exposed to previous oxidative stress.”
This observation and others come from experiments in which Brandl and coinvestigators exposed E. coli O157:H7 for either 15 minutes or 30 minutes to a juice made from crushed, liquefied leaves of fresh romaine lettuce to mimic the chemical compounds that are leaked from plant cells when lettuce is injured. An approach known as “microarray-based whole genome transcriptional profiling” enabled the researchers to determine which E. coli genes were activated.
“The technology gives us a snapshot or quick overview of all of the genes that were in play at those points in time,” says Brandl. “It’s an excellent technology for spying on the pathogen and learning about what happens to the pathogen at the molecular and chemical level.”
Study Is First To Provide Extensive Details
The microarray-based study was the “first to provide extensive information about the biology of E. coli O157:H7 in fresh-cut lettuce,” according to Brandl. “We showed that E. coli adapts well, using its genetic arsenal to protect itself against a multitude of assaults, including oxidative stress, osmotic stress, damage to its DNA, antimicrobial compounds exuded by the plant leaves, and other threats to its ability to survive and multiply. We showed that E. coli can adapt quickly. We also showed that E. coli exposed to the contents of broken lettuce cells activated genes that are associated with other key traits.”
|
|
Those traits included virulence, motility (the microbe’s ability to propel itself with its flagella), and its ability to attach to surfaces using appendages known as “fimbriae.”
“From what we’re observing with the microarray analyses,” Brandl says, “we hope to help develop new technologies that can overcome E. coli defenses. The microarray technology gives us an inside look at the numerous stresses that E. coli faces at the cut surface of a lettuce leaf. Each stress is a natural obstacle that E. coli has to overcome. We might be able to use these obstacles in a ‘hurdle’ approach to decontamination. Instead of relying on just one procedure or strategy, hurdle technology combines several strategies, each enhancing the other to weaken and kill the pathogen.”
Brandl and Albany colleagues Jennifer L. Kyle, Craig T. Parker, and Danielle Goudeau published their findings in Applied and Environmental Microbiology in 2010.—By Marcia Wood, Agricultural Research Service Information Staff.
This research supports the USDA priority of ensuring food safety and is part of Food Safety, an ARS national program (#108) described at www.nps.ars.usda.gov.
Maria T. Brandl is in the USDA-ARS Produce Safety and Microbiology Research Unit, Western Regional Research Center, 800 Buchanan St., Albany, CA 94710; (510) 559-5885.
"Leafy Greens: Keeping Salad Favorites Safe To Eat" was published in the April 2011 issue of Agricultural Research magazine.