Flu Fighters
Identifying, Treating, and Preventing
the Flu With Help From ARS Research
|
|
The first reports of a novel swine-origin influenza outbreak surfaced in the U.S. media in April 2009. The U.S. Centers for Disease Control and Prevention (CDC) first detected human infection in mid-April. Over the next 6 weeks, human infections were detected in more than 40 countries and nearly every U.S. state.
Early media reports referred to the virus—now known as “2009 influenza A (H1N1)”—as “swine flu,” a potentially confusing term for the public. Scientists initially concluded that the virus came from pigs because its genetic material was most similar to that of swine influenza virus. We now know that the 2009 H1N1 virus is a “triple reassortant virus,” meaning that it contains genetic material from swine, avian, and human influenza viruses—a mix that may help the virus spread quickly and pass between humans and pigs.
For some researchers, the story of a new influenza virus infecting pigs and people was all too familiar. As early as 2007, researchers from USDA’s Agricultural Research Service (ARS) and Animal and Plant Health Inspection Service (APHIS) had been monitoring a strain of influenza that could spread between pig and human populations.
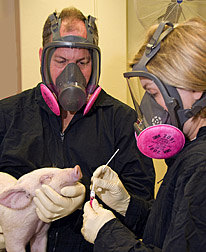
In August 2007, several people developed flulike symptoms after exhibiting their swine at a county fair in Ohio. Many of the pigs became sick as well. Tests revealed the source of the illness: a triple reassortant influenza A (H1N1) very similar to viruses that are endemic in U.S. pig herds.
|
|
Scientists from ARS and APHIS characterized the virus and found that in pigs it was slightly more virulent than average. In light of the virus’s characteristics and its documented ability to spread from pigs to humans, the scientists advocated close monitoring of influenza in swine, birds, and other species.
In September 2008, ARS, APHIS, and CDC launched a collaborative effort funded by CDC to develop a national swine influenza virus (SIV) surveillance pilot program. The goal is to better understand the epidemiology of SIV infections and to improve diagnostic tests, preventive management, and vaccines for swine and humans. This program has been instrumental in implementing surveillance for the 2009 outbreak, but it’s not the first time ARS influenza work has had major benefits. ARS scientists have been at the forefront of avian and swine influenza research for decades.
Agency scientists have developed tests to rapidly identify poultry infected with avian influenza and vaccines to protect against the disease. They’ve also developed and tested vaccines for swine. And ARS scientists have contributed to public health by identifying flu strains, assessing vaccines, and developing international standards for inactivating flu virus in cooked products.
Swine Surveillance
A 2008 paper co-written by ARS veterinary medical officer Amy Vincent states that swine influenza has undergone dramatic changes in the past decade, after nearly 80 years of relative stability. The increase in new subtypes and variants has complicated efforts to control the disease and increased the need for innovative strategies to fight it.
One such strategy is the pilot SIV surveillance program, in which APHIS coordinates with the National Animal Health Laboratory Network to screen potential SIV cases submitted from veterinary diagnostic laboratories. ARS then characterizes virus samples of special interest. The agencies also share virus isolates.
“We hope the pilot program will eventually become a permanent fixture,” Vincent says. “Having an established program would enable us to track not only where SIV occurs, but whether the virus undergoes significant mutations. This information could be essential in the event of an outbreak.”
The virus behind the current outbreak—like the virus from the Ohio county fair—is a type of influenza A known as “H1N1.” Though these two viruses have some substantial genetic differences, they both have surface proteins from swine influenza viruses. The surface proteins of these viruses are very different from those of human seasonal influenza A (H1N1) viruses, which means that most humans do not have any cross-reactive antibody against them. Other viruses that have caused human illness, but originated in animals, include H5N1 and H7 viruses from poultry.
At the beginning of the 2009 H1N1 outbreak in humans, pork producers had two important questions: Is the virus capable of infecting pigs? And if so, how will they respond? To find out, Vincent and her colleagues challenged young pigs with the virus in a biosafety level 3 facility at the ARS National Animal Disease Center (NADC) in Ames, Iowa. They observed that the pigs developed clinical signs of influenza consistent with those seen in endemic SIV infections. The scientists are also examining antisera and existing vaccines to determine whether the immunity that develops in response to infection or vaccination will protect pigs against the current outbreak.
Pigs, Poultry, and Pathogenicity
Because the new 2009 H1N1 virus contains genetic material from avian influenza viruses, two more questions must still be addressed: Is the virus capable of infecting avian species? And are poultry and other birds potential vectors for influenza in humans? Though the answers to these questions are still unknown, ARS scientists have a strong foundation of avian influenza research on which to build.
Scientists at the ARS Southeast Poultry Research Laboratory (SEPRL) in Athens, Georgia, have developed and evaluated avian influenza vaccines, helped assess public health threats, evaluated virus virulence, and helped develop protocols for inactivating flu viruses in food products. More recently, they’ve been evaluating how specific viruses pass from one animal to another.
Domestic pigs generally catch influenza from other pigs, but they’re also susceptible to infection from humans and birds. Fortunately, several highly pathogenic strains of avian influenza virus don’t appear to cause significant disease in pigs. That’s the conclusion of a study led by Aleksandr Lipatov (now with the CDC). The research team also included Yong Kuk Kwon, Luciana Sarmento, Erica Spackman, David Suarez, and David Swayne at SEPRL, and Kelly Lager at NADC.
“The finding is significant because pigs are thought to be vessels for mixing avian influenza and human influenza to allow genetic reassortment that could set the stage for pandemic avian influenza,” says Swayne.
In the studies, piglets were exposed to four different strains of highly pathogenic H5N1 avian influenza viruses. Two swine influenza viruses were used as controls. The studies show that none of the H5N1 virus strains caused significant or fatal disease in pigs, but they did cause mild to moderate inflammation in the lungs.
Infection with either swine influenza virus resulted in much more severe symptoms and pneumonia.
In related research, scientists at NADC and SEPRL have collaborated to develop two sets of diagnostic tests to quickly and accurately differentiate the new 2009 H1N1 virus from endemic forms of swine and avian influenza viruses already circulating in the United States.
Research efforts like these are essential to ensuring the health and safety of people and livestock in the United States and around the world.—By Laura McGinnis and Sharon Durham, Agricultural Research Service Information Staff.
This research is part of Animal Health, an ARS national program (#103) described on the World Wide Web at www.nps.ars.usda.gov.
To reach scientists mentioned in this article, contact Laura McGinnis, USDA-ARS Information Staff, 5601 Sunnyside Ave., Beltsville, MD 20705-5129; phone (301) 504-1654, fax (301) 504-1486.
"Flu Fighters: Identifying, Treating, and Preventing the Flu With Help From ARS Research" was published in the August 2009 issue of Agricultural Research magazine.