Glomalin What Is It . . . and What Does It Do?
Glomalin is a glycoprotein, a sugar-protein compound that might trigger the formation of soil.
In a study at two Mandan, North Dakota, locations, Kristine Nichols, a microbiologist with the ARS Northern Great Plains Research Laboratory, found that soils under native grasses—switchgrass, blue grama, big bluestem, and indiangrass—have higher levels of glomalin than soils planted to nonnative grasses, such as Russian wildrye, intermediate wheatgrass, crested wheatgrass, and western wheatgrass.
“The more glomalin in a particular soil, the better that soil probably is,” Nichols says.
Native Grasses Mean More Glomalin
In 2004, Nichols collected soil under monoculture grass plots established between 1987 and 2002. She found that the amount of glomalin in the soil increased as the degree of interdependence increased between plants and arbuscular mycorrhizal fungi. These fungi produce glomalin and live inside plant roots and in the surrounding soil. The fungi have hairlike filaments called “hyphae” that extend the reach of plant roots. The plant-fungus interdependence is greatest in warm-season native grasses, such as switchgrass, blue grama, big bluestem, and indiangrass.
Further evidence that soils under native grasses are higher in glomalin came from another study on rangeland areas at Mandan and near Platte, South Dakota. This study also showed that shifting cattle to other pastures before they could overgraze an area helped raise glomalin levels in soils.
In an earlier study, Nichols analyzed samples from undisturbed soils under native vegetation in Maryland, Georgia, and Colorado. According to her analysis, glomalin stored an average of 15 percent of the carbon found in those soils and contributed greatly to soil fertility. The highest amount of stored carbon, 30 percent, was found in a Colorado soil and the lowest amount, 9 percent, in a Georgia soil. These amounts are similar to those from other soil samples taken around the world.
Glomalin may also provide a secure vault for soil carbon. By helping to form and stabilize small soil particles called “aggregates,” glomalin reduces the decomposition of the labile (unstable) organic carbon compounds within the aggregates. For this reason, the carbon within the glomalin molecule may resist decomposition for up to 100 years.
The larger amounts of glomalin and stored carbon observed under grasses such as switchgrass add to the grasses’ value as potential sources of cellulosic ethanol.
A Sticky String Bag
Nichols began her career with ARS working with soil scientist Sara Wright, who discovered and named glomalin in 1996. Wright has since retired.
Nichols explains how glomalin may actually make soil.
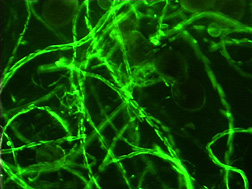
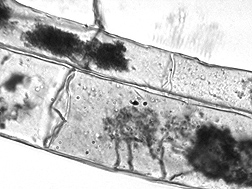
“Before the discovery of glomalin, other researchers described the hyphae of mycorrhizal fungi as forming a sticky string bag, with the hyphae acting as the string and some other substance on the hyphae sticking sand, silt, and clay particles, plant debris, and other organic matter to the hyphae—like little gobs of chewing gum. Photographs of glomalin on hyphae indicate that glomalin might be these ‘gobs of chewing gum.’ The sticky string bag starts aggregate formation, which is a major part of what makes soil. Aggregates provide structure to soil for better water infiltration, water-holding capacity, and gas exchange, and increase soil fertility by providing organic carbon (that is, food) to soil organisms, which use this food as energy to release plant nutrients from the soil,” Nichols says.
“On the surface of soil aggregates, glomalin no longer acts like bits of chewing gum, but instead forms a latticelike waxy coating to keep water from flowing rapidly into the soil aggregate and causing the aggregate to rupture. When an aggregate ruptures, everything within it—plant debris, organic matter, microbes, glomalin, fungal hyphae, and sand, silt, and clay particles—may be washed or blown off the soil with the erosive forces of wind and water,” Nichols explains.
The waxy nature of glomalin also serves as a protective coating on fungal hyphae, keeping water and nutrients from being lost before they reach the plant host and protecting hyphae from decomposition and microbial attack. It may also be what gives hyphae the rigidity needed to span air spaces between soil particles and extend beyond plant roots for greater access to more plant nutrients present in the soil.
Nichols uses glomalin measurements as a quick guide to evaluate how soil friendly farming or rangeland practices actually are. The amount of glomalin present is also a measure of how much carbon is being stored under various practices, so quantifying glomalin could be used in conjunction with carbon-credit-trading programs.
Nichols has done studies on cropland as well as on rangeland. On cropland, she found that both tillage and fallowing—as is common in arid regions such as those in the Northern Plains—lower glomalin levels by destroying living hyphal networks. The networks are physically torn by tillage or broken down due to starvation during fallowing.
The More Glomalin, the Better Soil’s Feel
The higher a soil’s glomalin level, the better its tilth, or feel and structure, the less its susceptibility to erosion by either wind or water, and the better it is for the growing plants.
As the builder of the formation bag for soil, glomalin is vital everywhere to soil building, productivity, and sustainability—as well as to carbon storage.
This is particularly true in the Northern Plains region of the United States, where rangeland covers 43 percent of the landscape. More than 60 percent of the soils on this land are highly vulnerable to wind erosion, which destroys soil and releases carbon dioxide to the atmosphere. The Northern Plains’ 180 million acres of rangeland represent a big chunk of the nation’s approximately 526 million acres of nonfederal rangeland.
All this makes glomalin both the rancher’s—and Earth’s—best friend.—By Don Comis, Agricultural Research Service Information Staff.
This research is part of Soil Resource Management, an ARS national program (#202) described on the World Wide Web at www.nps.ars.usda.gov.
Kristine Nichols is in the Natural Resource Management Research Unit, USDA-ARS Northern Great Plains Research Laboratory, 1701 10th Ave., SW, P.O. Box 459, Mandan, ND 58554-0459; phone (701) 667-3008, fax (701) 667-3054.
"Glomalin What Is It . . . and What Does It Do?" was published in the July 2008 issue of Agricultural Research magazine.