Inflamatory News about Fat Cells
|
|
Deep within bone marrow, spleen, and lymph nodes lie a form of large white blood cells. As part of the immune system, these “macrophages,” as they are called, circulate within blood to seek out and devour bacteria, other foreign invaders, and cellular debris.
Scientists have known that macrophages also appear in adipose, or fat, tissue. But their role in the tissue was once thought to be inconsequential. Now, a new study explains important aspects of these immune cells in body fat, such as why they infiltrate, how they function, and just where they gather. The study was authored by Andrew Greenberg, Martin Obin, and colleagues from Italy and Canada. Both scientists are with the Obesity and Metabolism Laboratory, which Greenberg heads, at the Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University in Boston, Massachusetts.
In recent years, scientists have looked at fat cells and their surrounding cells not just as yellow blobs that make our clothes too tight, but more as an organ, or neighboring bodies of organic fatty tissue. The fat cells, or “adipocytes,” within this tissue have several functions, such as pumping out vital energy-producing fatty acids, storing fatty acids for future use, and secreting hormones that regulate body weight. But unfortunately, among the obese, they’re a source of inflammatory chemicals.
|
|
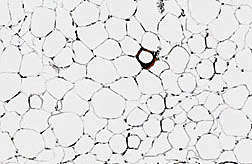
No matter how much one eats, fat cells appear to be limited in the size they can attain. The amounts of lipids that enter these fat cells gradually increase as people gain weight. Greenberg and his colleagues found that as fat cells reach their maximum size, they eventually break down and die. When obesity occurs and is maintained, new fat cells form to store the fat—but as those cells reach their peak size, they also eventually die.
Greenberg’s study shows that more than 90 percent of the macrophages in adipose tissue of obese mice and humans are located around dead fat cells. As the fat cells get bigger within fat tissue, the prevalence of macrophages increases proportionally.
Though recent research by other scientists has shown that macrophages are responsible for most of the inflammatory molecules released within adipose tissue, why they produce these chemicals is unknown.
The new findings may partly explain the mechanisms inside the adipose tissue that cause the cells to secrete the inflammatory chemicals.
Based on Greenberg’s study, these immune cells now appear to be rushing to fat cells after their death to mop them up. When this happens, the macrophages may emit potentially dangerous amounts of inflammatory chemicals.
“When fat cells die, macrophages surround the dead lipids the same way white cells surround a wooden splinter in your skin,” Greenberg says. “The immune system is essentially surrounding and sequestering the dead fat cells and gorging on the leftover lipids and cellular debris.” The macrophages identify the leftover insoluble lipid droplets as foreign bodies.
In a case of molecular rescue gone awry, the findings may explain how enlarged fat cells, as found in obesity, promote obesity-related complications such as arthritis, insulin resistance, diabetes, or heart disease.—By Rosalie Marion Bliss, Agricultural Research Service Information Staff.
This research is part of Human Nutrition, an ARS National Program (#107) described on the World Wide Web at www.nps.ars.usda.gov.
Andrew Greenberg is at the USDA-ARS Obesity and Metabolism Laboratory, Jean Mayer USDA Human Nutrition Research Center on Aging at Tufts University, 711 Washington St., Boston, MA 02111-1524; phone (617) 556-3144, fax (617) 556-3224.
"Inflamatory News about Fat Cells" was published in the March 2006 issue of Agricultural Research magazine.